Scientific & Clinical Evidence

NMP (Natural Matrix Protein) Technology is backed by extensive scientific and clinical data, which continues to prove the safety and efficacy of our products.
CLINICAL EVIDENCE
- 50 patients undergoing 1–2 level ALIF
- Treated with NMP alone (no local autograft or bone marrow aspirate)
- Medically complex population (98% with ≥1 comorbidity; 72% with ≥2)
- Fusion outcomes comparable to autograft
- 100% <3° motion on flexion-extension radiographs
- CT-confirmed bridging bone in 89.1% of levels (Grade 3A)
- Remaining levels demonstrated progressive bone formation without fixation failure
- Statistically and clinically significant improvements in pain and disability scores
Summary: NMP demonstrated fusion performance comparable to autogenous bone graft in a high-risk, real-world patient population.
- First 20 patients in an ongoing prospective cervical/lumbar fusion study (NCT06000319)
- 90% fusion rate at 12 months across lumbar interbody approaches
- Clinically meaningful improvements in pain and disability
- High-risk cohort with multiple comorbidities (diabetes, hypertension, obesity, smoking)
- No serious adverse events related to the investigational product
Summary: Early prospective results support NMP’s ability to promote fusion in medically complex spine patients.
Summary: Data from this study showed that NMP bioimplants demonstrated high fusion rates and a favorable safety profile clinically in ALIF procedures, with outcomes comparable to those reported for INFUSE in ALIF procedures.
CASE STUDIES
SPINE & ORTHO
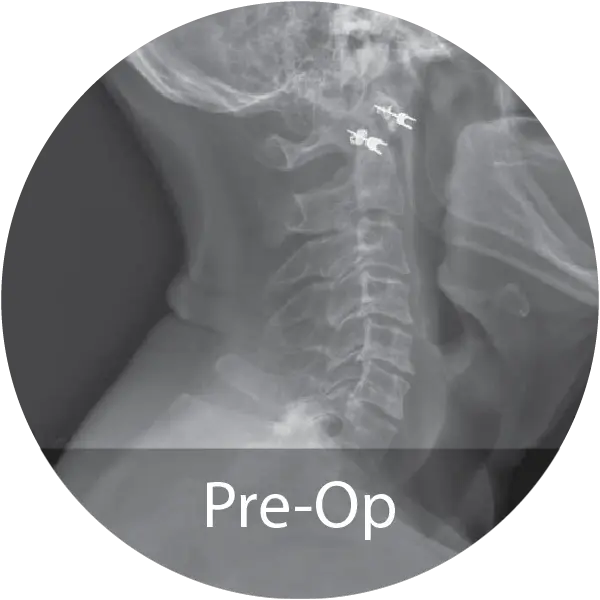
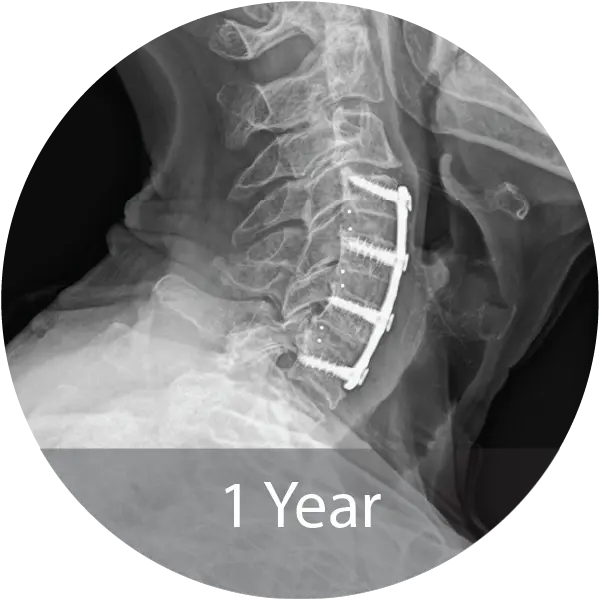
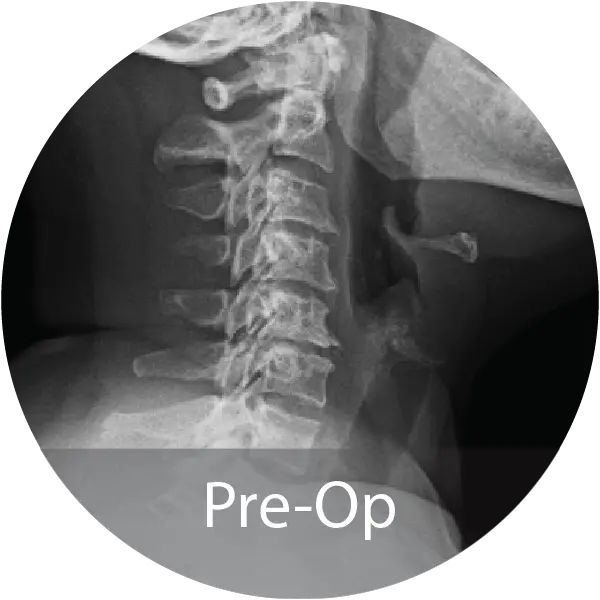
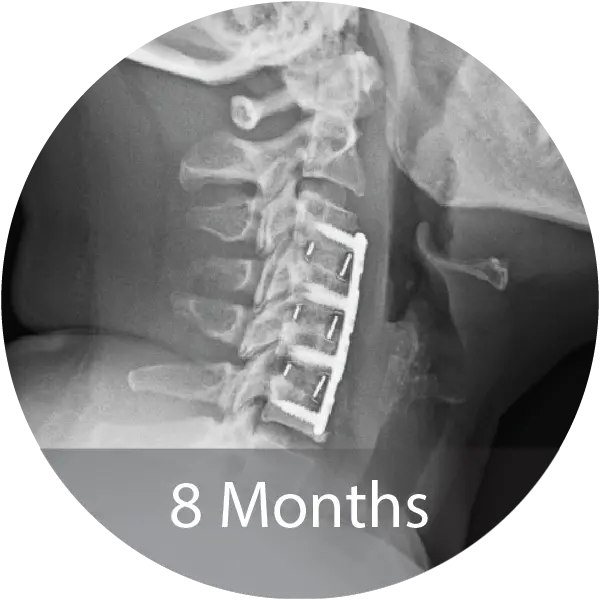
Case of an 80-year-old female with cervical degenerative disc disease and stenosis treated with a 3-level ACDF using NMP® Fibers. Reported 12-month fusion, no adverse events, complete pain resolution, and excellent handling characteristics.
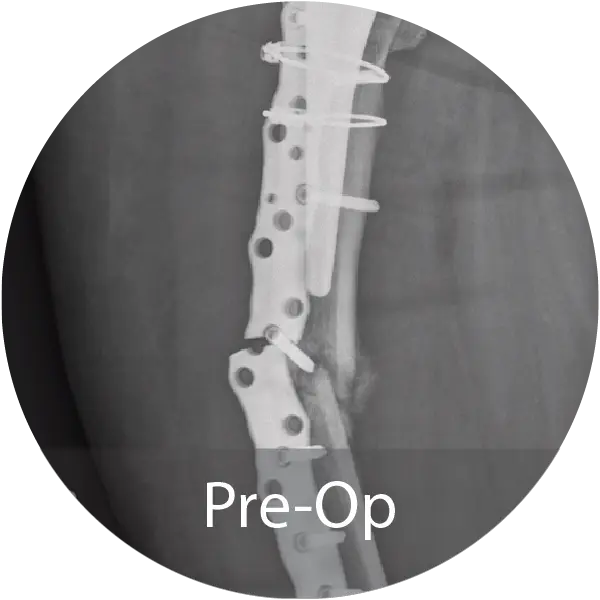
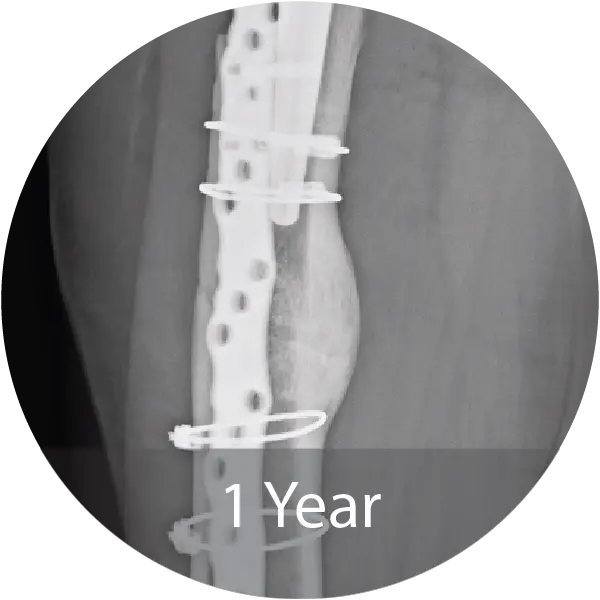
Case of an 81-year-old female with a periprosthetic femoral non-unionfracture and failed hardware treated with repeat ORIF and NMP fibers hydrated with saline. Progressive callus formation on x-ray and patient reported no pain and full range of motion 1 year post-op.
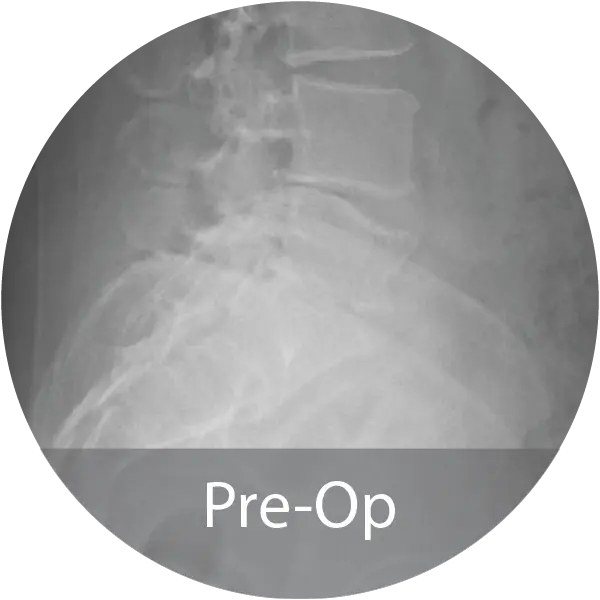
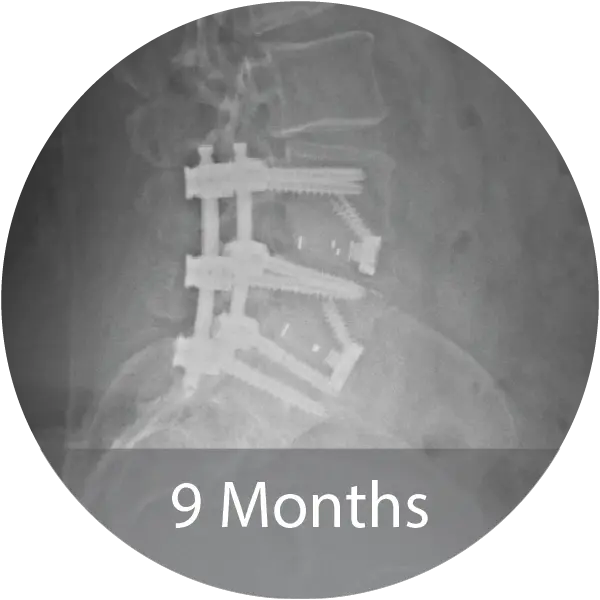
Two case series including a lumbar case (59-year-old female, 1 pack/day smoker) treated with L4–S1 ALIF with posterior fixation and a cervical case (60-year-old female, 1.5 pack/day smoker) treated with 3-level ACDF demonstrated solid arthrodesis by 9 months, significant improvements in pain and disability scores, no complications, and return to work. A cost analysis showed a 26–29% reduction in biologic cost per case when transitioning from INFUSE® and i-FACTOR® to NMP® Bioimplant, translating to an estimated annual savings exceeding $270,000 for the facility.
FOOT & ANKLE
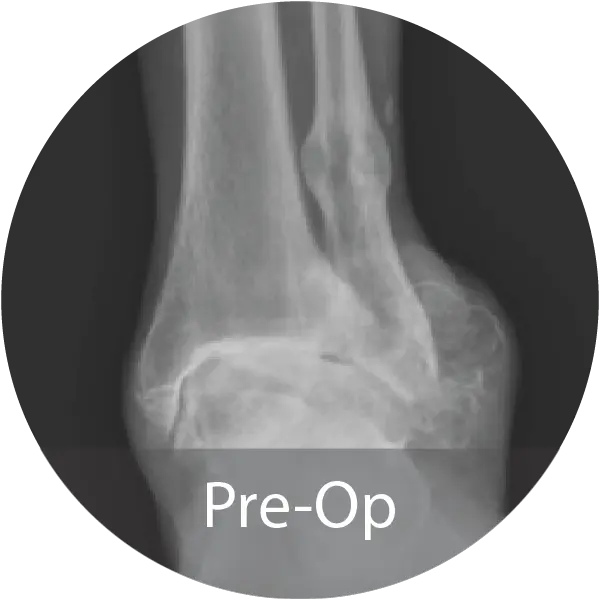
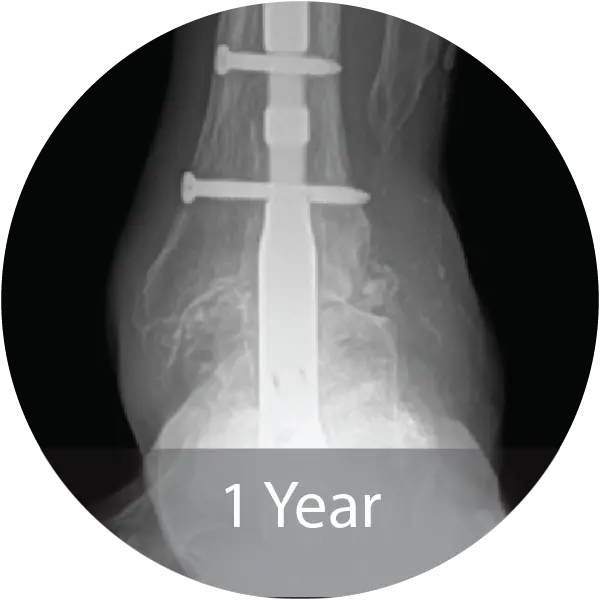
Case of 64-year-old female with hindfoot arthritis treated with tibiotalocalcaneal arthrodesis and intramedullary nailing using InduceXT®. Reported solid osseous consolidation with no postoperative complications, pain-free ambulation, and full return to activities.
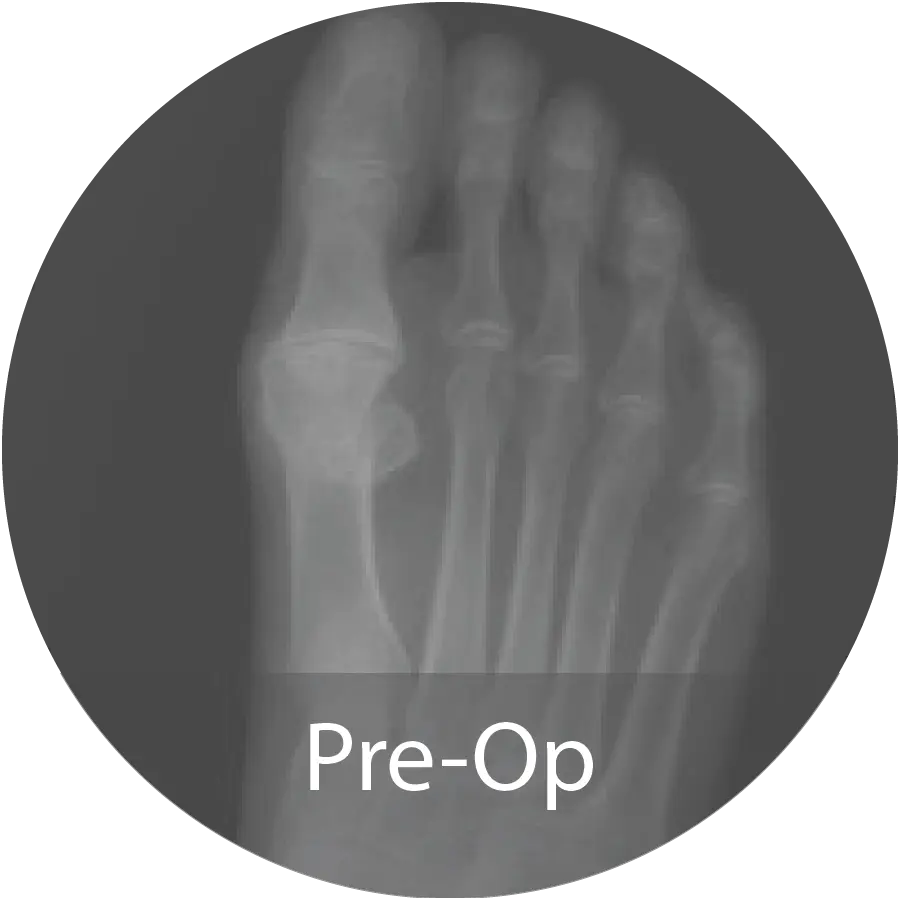
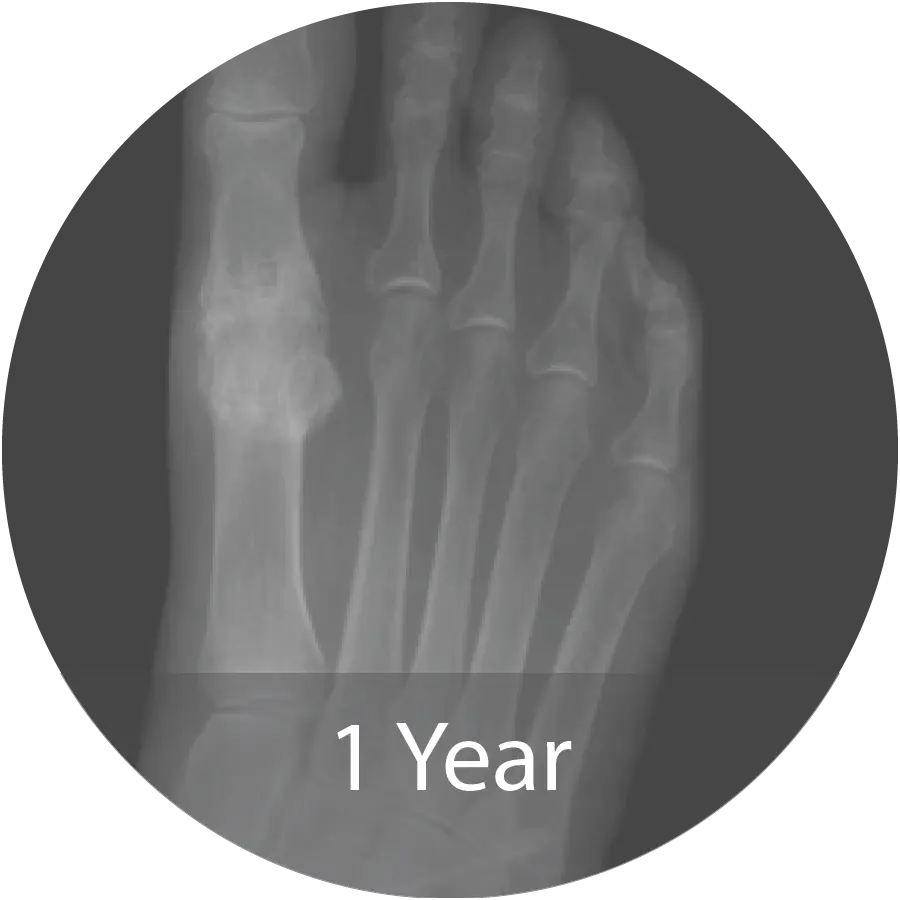
Case of 74-year-old female with hallux rigidus and multiple metallic allergies treated with 1st MTPJ arthrodesis using biointegrative hardware and InduceXT® hydrated with normal saline. Reported complete osseous consolidation at 12 months and pain-free outcome.
Case of 68-year-old female with post-traumatic osteoarthritis treated with subtalar joint arthrodesis using InduceXT®. Reported successful fusion at 12 months with no postoperative complications, pain-free ambulation, and return to normal activities.
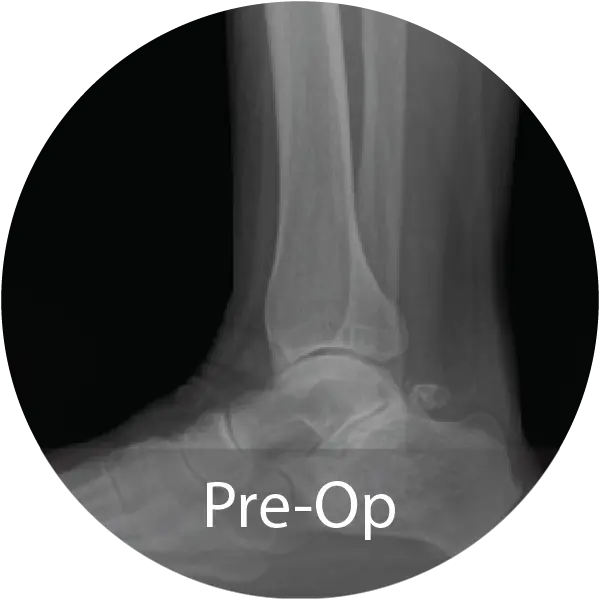
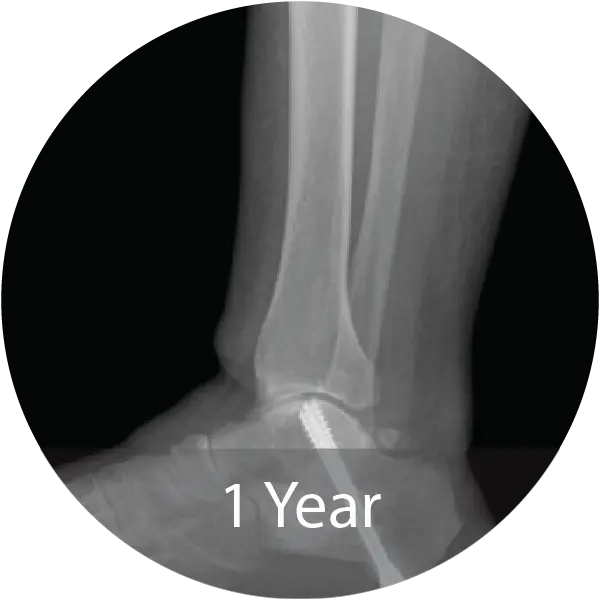
Case of 79-year-old male with progressive collapsing foot deformity and secondary ankle osteoarthritis treated with triple arthrodesis and total ankle replacement using InduceXT®. Reported solid osseous fusion with well aligned hindfoot and ankle, no pain during ambulation, and full return to activities.
DENTAL
This 69-year-old male presented for extraction of tooth #4 for a failing root canal with decay. An alveolar ridge preservation procedure was performed utilizing INDUCE Oi-9®.
Fiber Matrix covered with an amnion chorion membrane.
Patient presented with complete healing at 5 months and implant placement took place.
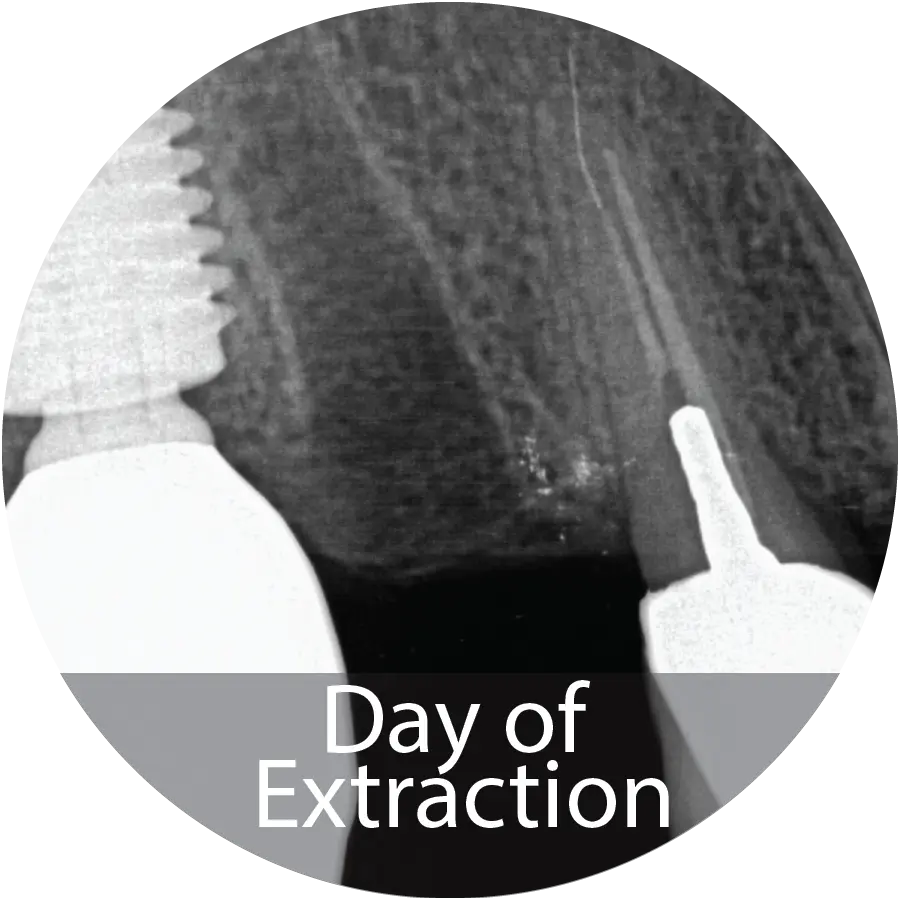
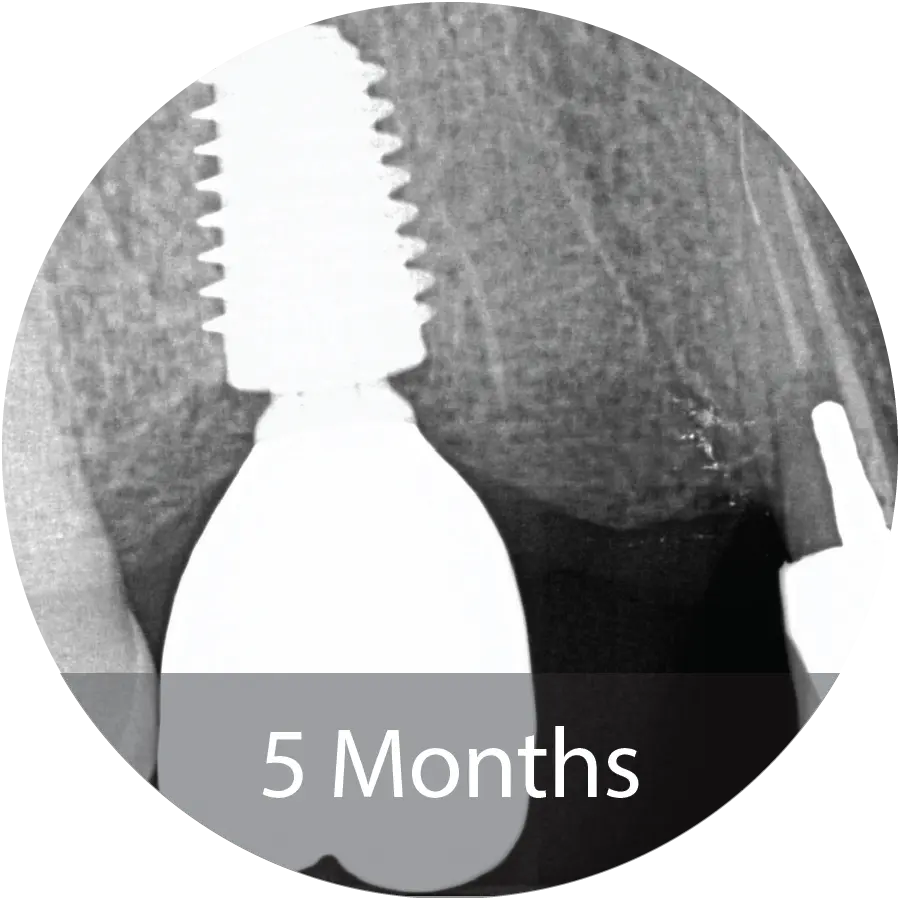
67-year-old male presented for extraction of tooth #18. An Alveolar ridge preservation procedure was performed utilizing INDUCE Oi-9® Fiber Matrix covered with an amnion chorion membrane.
Evaluation of the bone core with micro-CT and histological staining demonstrated mineralized tissue consistent with new bone, surrounded by loose trabecular structures. Implant placement was performed at 6 months following grafting.
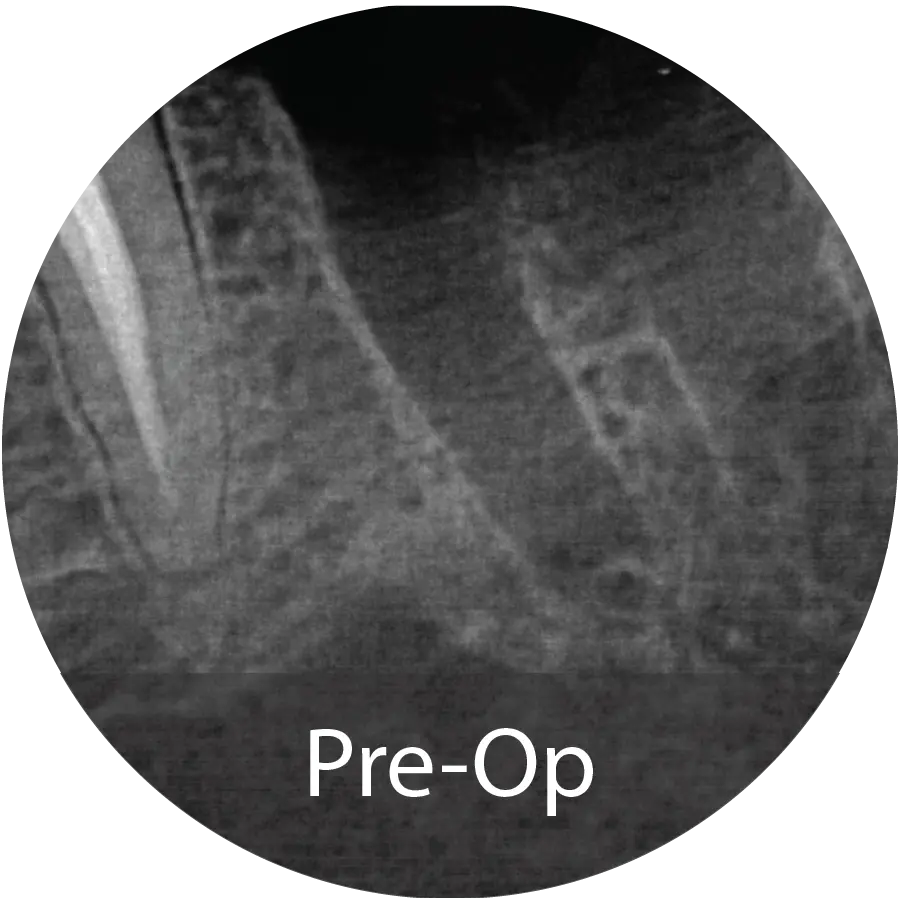
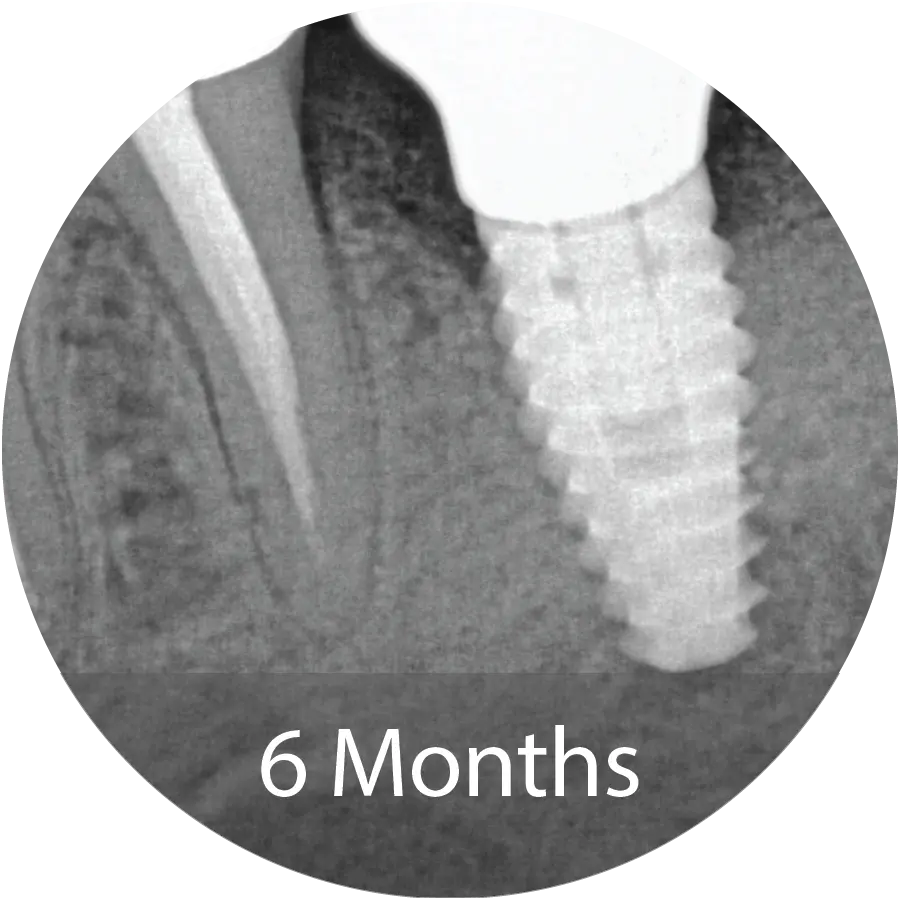
This 88-year-old female presented for extraction of tooth #20. Following the extraction, alveolar ridge preservation procedure was performed utilizing INDUCE Oi-9® Fiber Matrix covered with an amnion chorion membrane.
This patient demonstrated complete bone regeneration at 6 months and implant placement took place.
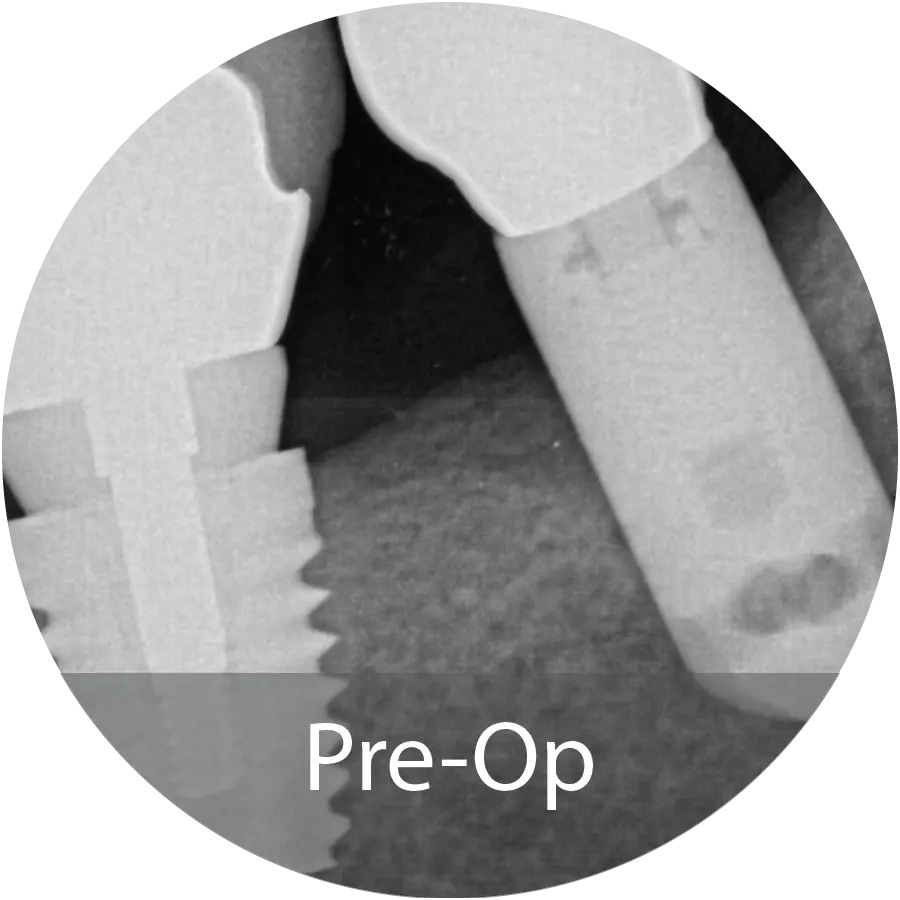
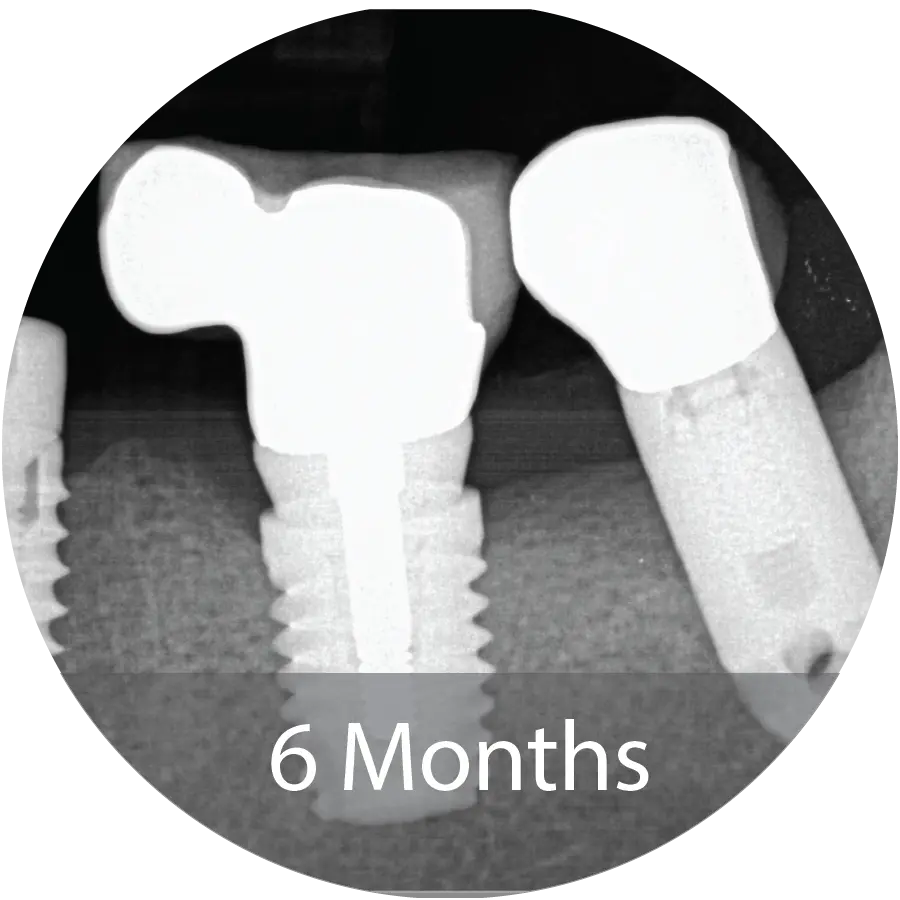
A healthy 73 year-old female had her anterior maxillary implants fail in 2011. She has since been wearing a partial anterior maxillary prosthesis. Recently, her maxillary right canine fractured, making the denture uncomfortable to wear. Her desire was to return to a dental implant supported restoration.
Excellent reconstruction of both vertical height and horizontal width was achieved following INDUCE Oi-9® augmentation. A titanium-reinforced growth factor membrane was used to maintain space during the healing process. CT post-operative horizontal measurement shows significant augmentation.
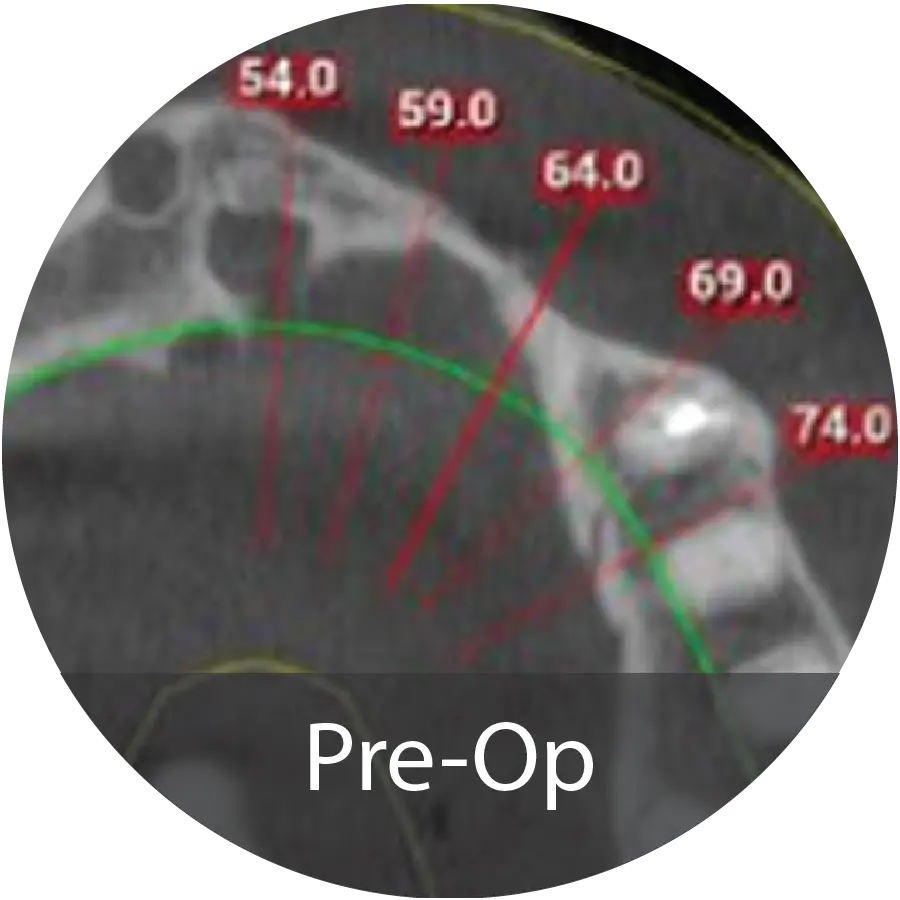
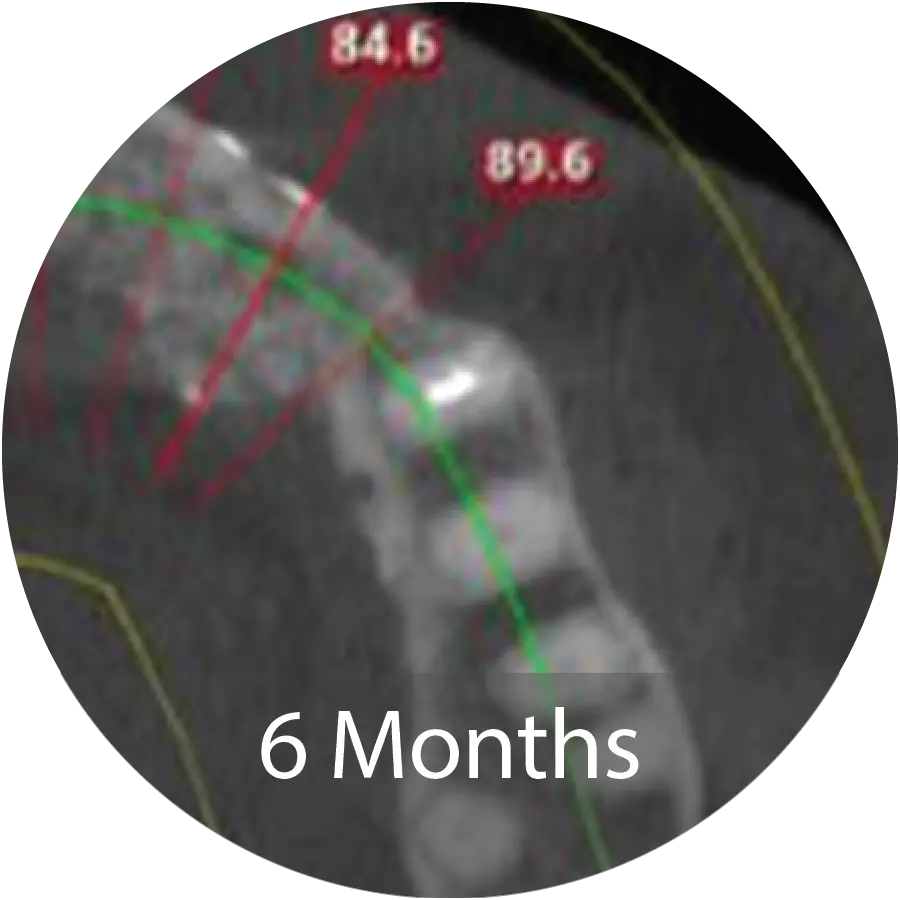
A 28-year-old Caucasian male with a history of long-term drug abuse and no other significant medical conditions presented for dental treatment for a maxillary overdenture supported with locator attachments. The patient had significant bone loss and insufficient support for dental implants.
The maxillary ridge was reconstructed using INDUCE Oi-9® Fiber Matrix with a perforated titanium reinforced PTFE membrane. At 6 months post operative CT scans show substantial new bone regeneration and ready for dental implant therapy.
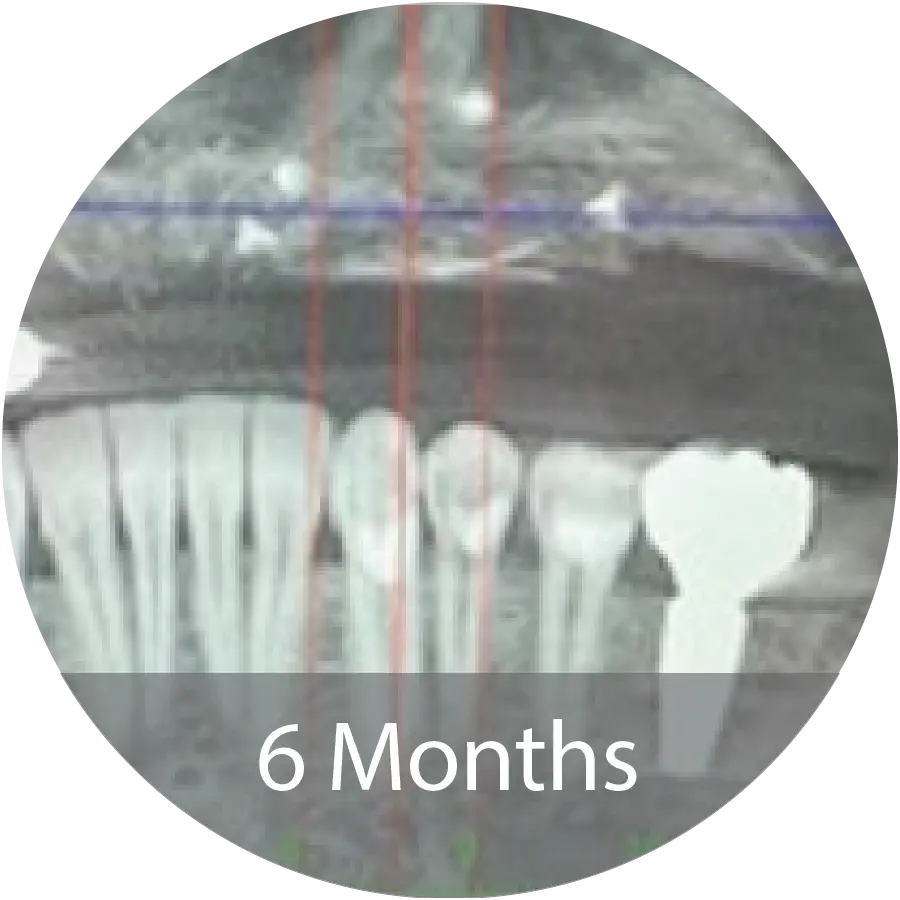
CLINICAL STUDIES (ONGOING & REGISTERED)
ANIMAL & SCIENTIFIC STUDIES
- Describes the development, processing, and biologic rationale for enhanced growth factor bioavailability
- The NMP® process forms BMP/NCP complexes and enhances BMP bioavailability
- NMP demonstrates osteoinductive potential in C2C12 in vitro assay
- NMP is osteoinductive in vivo
- NMP generates both an early burst release of BMPs followed by a prolonged release phase over 10+ weeks
- Evaluates comparative biologic performance of NMP relative to commercially available grafts in a rat spine fusion model
- NMP induced robust, organized trabecular bone with marrow-like regions
- Greater bone volume and quality observed with NMP as compared to BMP-2
Link Coming Soon
Proprietary processing increased bioavailability of key growth factors: BMP-2, BMP-7, VEGF, PDGF-BB, and TGF-β1
- NMP stimulated osteoinductive activity in-vitro (C2C12 alkaline phosphatase assay)
- In-vivo testing in the FDA-recognized athymic rat muscle pouch model: NMP formed significantly more mineralized tissue than DBM or INFUSE™ and bone was distributed throughout the implant rather than a peripheral “shell”
- Compared NMP to commercially available cellular, peptide, and growth factor-enhanced grafts
- Athymic rat muscle pouch model
- NMP consistently produced new bone throughout the implant
- INFUSE™ produced less bone with cyst-like ossicle formation
- OsteoAMP® and iFactor® did not demonstrate osteoinductive activity
- Cellular graft performance was variable and preparation-intensive